About Purixan® Test
Indication
PURIXAN (mercaptopurine) is indicated for the treatment of patients with acute lymphoblastic leukemia as part of a combination regimen.
Liquid Formulation
PURIXAN offers patients and healthcare professionals a liquid formulation as an alternative to the mercaptopurine tablet.
- Mercaptopurine is a cornerstone of all maintenance therapy drug regimens for acute lymphoblastic leukemia (ALL).1
- The efficacy of mercaptopurine in keeping children and adults with ALL disease free has been established by a number of national and international trials.1
- Prolonged treatment with mercaptopurine is proven to increase disease-free survival.1
- Until recently, the only marketed formulation of mercaptopurine has been a 50-mg tablet.
- However, solid oral dosage forms may be ineffective for many children because they have difficulty swallowing tablets and capsules.2
- Fixed doses are impractical because the dose varies according to the size of the child.2
- More than 90% of pediatricians reported that a drug’s taste and palatability were the greatest barriers to compliant treatment.2
- For children, PURIXAN addresses common concerns relating to treatment with a tablet.
Dosing and Administration
Combination Chemotherapy Maintenance Regimens with PURIXAN
- Recommended Starting Dose: 1.5 to 2.5 mg/kg (50 to 75 mg/m2) as a single daily dose.
- Monitoring: After initiation of treatment with PURIXAN, continuation of appropriate dosing requires periodic monitoring of the patient’s absolute neutrophil count (ANC) and platelet count to assure sufficient drug exposure (that is to maintain ANC at a desirable level) and to adjust for excessive hematological toxicity.
- Thiopurine S-Methyltransferase (TPMT)–Deficient Patients
- Dosage reduction is generally required in patients with inherited little or no TPMT activity, due to increased risk for severe mercaptopurine toxicity.
- Testing for TPMT gene polymorphism should be considered in patients who experience severe bone marrow toxicities.
- Homozygous TPMT-deficient patients may require up to a 90% dose reduction (10% of the standard PURIXAN dose).
- Most patients with heterozygous TPMT deficiency tolerated recommended mercaptopurine doses, but some required dose reduction based on toxicities.
- Dosage Forms and Strength: Oral suspension: 2000 mg/100 mL (20 mg/mL); pink to brown in color.
Comparison between PURIXAN and the tablet form of mercaptopurine
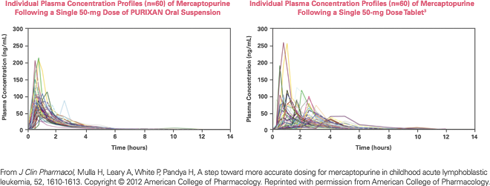
The bioavailability of PURIXAN is equivalent to that of the tablet form of mercaptopurine, as measured by the AUC (area under the concentration curve).
- The mean ratio of the AUCs for PURIXAN and mercaptopurine tablets is 114%, with a 90% confidence interval of 108%–121%. 3
- The accepted target range of AUC ratios for equivalence is 80%–125%.3
Consistent Absorption
PURIXAN reduced the variability in the absorption of mercaptopurine, proving itself to be a dependable and reliable alternative to the tablet.3
Predictability
PURIXAN performs more consistently and predictably than the mercaptopurine tablet.
- PURIXAN had substantially lower between-subject variability in maximum plasma concentrations (46% vs 69% coefficient of variation).3
Flexibility and Accuracy
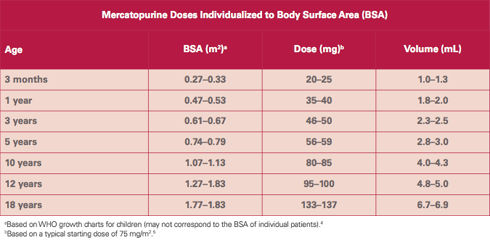
PURIXAN provides dosing flexibility and accuracy.
PURIXAN 20 mg/mL oral suspension provides an accuracy of 2 mg and an opportunity to administer the product in a way that may be more acceptable to children.2
- Pui CH, Evans WE. Treatment of acute lymphoblastic leukemia. N Engl J Med. 2006;354:166-178.
- Mennella JA, Spector AC, Reed DR, Coldwell SE. The bad taste of medicines: overview of basic research on bitter taste. Clin There. 2013;35:1225-1246.
- Mulla H, Leary A, White P, Pandya H. A step toward more accurate dosing for mercaptopurine in childhood acute lymphoblastic leukemia. J Clin Pharmacol. 2012;52:1610-1613.
- WHO growth chart for children. http://www.who.int/childgrowth/standards/en/. Accessed April 24, 2014.
- PURIXAN (mercaptopurine) [package insert]. Franklin, TN: Rare Disease Therapeutics, Inc.; May 2017.